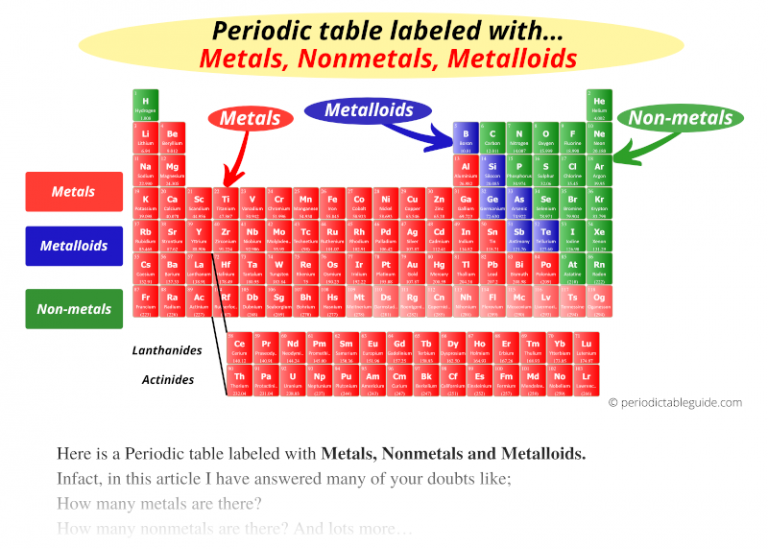
Elements in a family have similar but not identical properties. Chemistry Portal v t e The chemical elements can be broadly divided into metals, metalloids, and nonmetals according to their shared physical and chemical properties.Each column of elements is called a family or group.Purple has 8 electrons in its outer shell.As you keep counting the colored columns, you add an additional electron. 
Elements in the orange group have 2 electrons in their outer shell.There results will be color coded on a periodic table (e.g. Elements in the red group have 1 electron in their outer shell. From your data, you will classify each element as a metal, a non-metal or a metalloid.Elements in the third period have 3 electron shells.Every element in the second period has two energy levels available.Every element in the top row (first period) has one orbital for its electrons.Metalloids have properties of both metals and nonmetals. The dividing line (zigzagline) between the metals and nonmetals are elements called semiconductors or metalloids. Properties include brittle, dull, not good conductors (insulators). The nonmetals are on the right side of the table. Properties include good conductors, shiny luster, malleable, ductile. In general, elements located in the left two-thirds or so of the periodic table are metals. 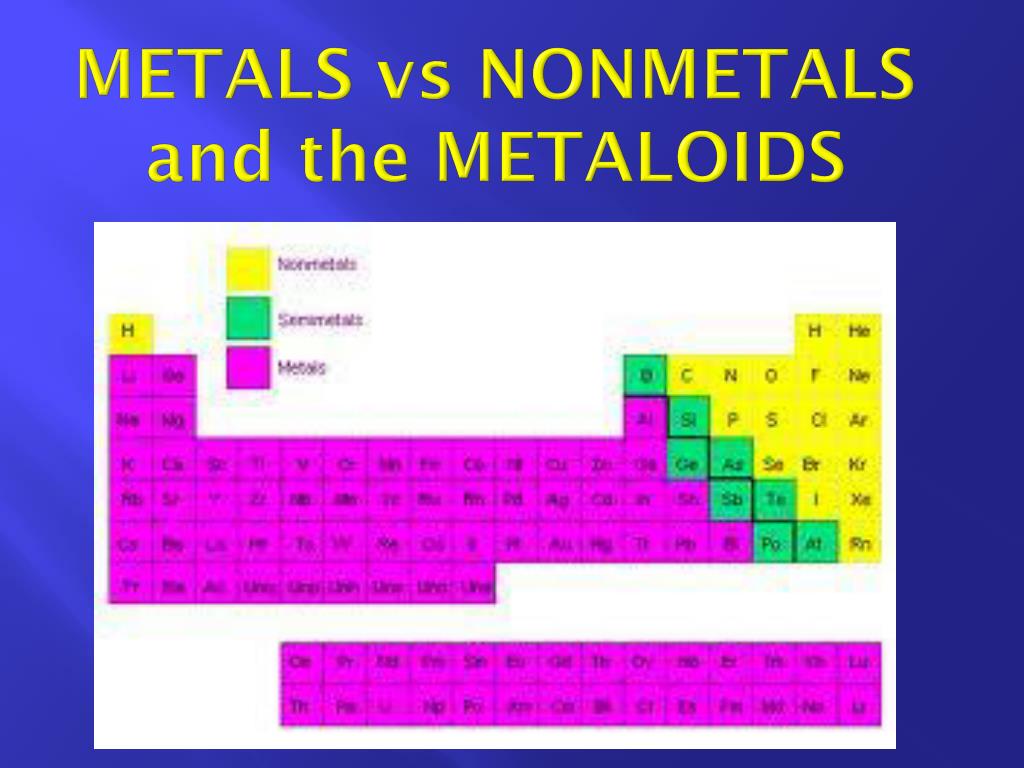
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
